It is estimated that 230,000 Australians are infected with Hepatitis C virus. In Cambodia the estimation is somewhere between 300,000 to 750,000 people. In late 2015 a number of new Direct Acting Antiviral drugs (DAAs) which cure Hepatitis C in 12 to 24 weeks with few side effects, became available via at least three different drug companies. Drug prices ranged from US$84,000 per 12 week course, forcing governments in high income countries to put strategies in place to offer treatments to their citizens.
In the UK where it is estimated 214,000 people are infected with Hepatitis C, the government capped the number of people able to be treated because of cost, to 20,000 people over a two year period. Most European governments made treatment only available to those with advanced liver disease. Australia took a different stance, negotiating with drug companies for a volume based price agreement. Treating a large number of people drove the cost per treatment down whilst ensuring profits to the drug companies. The government assured DAA treatment to all Hepatitis C infected citizens free of charge, an ambitious and dramatic move which I was excited to be a part of for a short but fulfilling time.
On the other hand, low to middle income countries have little to no chance of offering treatment to their citizens outside of the private market, which is susceptible to all kinds of errors. In Australia patients attend an appropriate clinic where they are determined as eligible for treatment. The few criteria are that you must be infected with a genotype of the virus that is treatable and be well placed and agree to good adherence of both treatment and follow up appointments. The registered clinician calls a ‘hotline’ where the patient’s identification and details are recorded on a centralised system alongside the prescribing doctor’s details and an approval number is given for the drug to be released by a registered pharmacy. In Cambodia, the drugs are available for a price, at private pharmacies without regulation. DAA treatment is only effective if the correct dose of two molecules (ie a combination of two drugs) are given together, for the correct duration, all of which requires clinical assessment following recommended guidelines. The private market side steps such structure, allowing for many errors including the risk of resistant strains of virus evolving when exposed to incorrect or incomplete treatments.
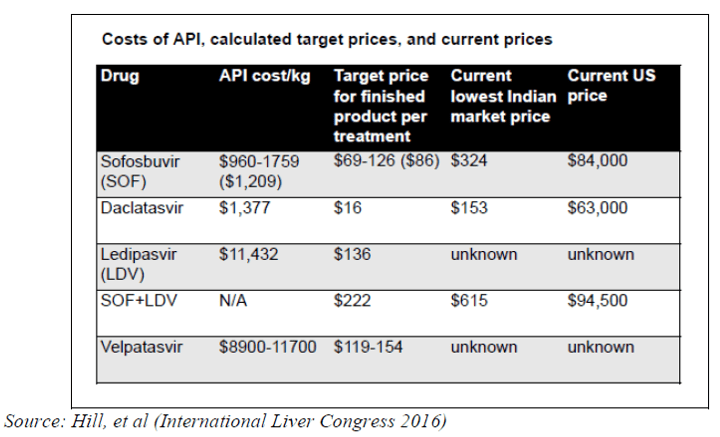
An example of the pricing game that pharmaceutical companies play is in the table below where API = Active Pharmaceutical Ingredient. The mark-up obviously makes a small number of people extremely wealthy.
The point of the MSF Hepatitis C Treatment program here in Cambodia is:
- to offer treatments to a population who would otherwise not be able to access such medications;
- to offer treatments to a small portion of the population who may be able to afford medications from the private market but may not receive the correct molecules for the correct duration to ensure cure (although there are clinicians who are qualified to offer appropriate care and treatment – but there is no regulation);
- to put strategies in place which will ultimately allow the Ministry of Health to take over a functioning HCV Treatment program;
- to advocate for reasonable treatment costs by negotiating with drug companies and/or introducing generic treatments which can be provided at a fraction of the patented costs.
 Gilead launched Sofosbuvir in the US at $1,000 per pill, although it can be mass produced for less than $1.00 per pill.
Gilead launched Sofosbuvir in the US at $1,000 per pill, although it can be mass produced for less than $1.00 per pill.
Some of my information herein comes from an organisation founded by Medecins sans Frontieres in partnership with six other organisations, and who we continue to work alongside: Drugs for Neglected Diseases Initiative (DNDi) . One of the best things about working with an international organisation is the exposure we get to global health care programs working effectively towards a more just world.

“Gilead launched Sofosbuvir in the US at $1,000 per pill, although it can be mass produced for less than $1.00 per pill.” 😦
LikeLike
There is a lot of immorality in big business, the Pharmaceuticals are right there among the worst. They have the potential to save millions from illness & death, but the $$ come first.
LikeLike